We have provided you with the best chemistry class 10 chapter 2 notes for federal board. We also have discussed the important questions as well which have been repeated in the board exams multiple times. We also have tried our best to deeply explain each question and clear the concepts of students. If students find difficulty in learning through these notes then they can visit our YouTube Channel for learning through visual aids.
Acid Base and Salts
Table of Contents
Chemistry Class 10 Chapter 2 Short Answer
1: Describe some important properties of acid and bases.
PROPERTIES OF ACID:
- Acids have sour taste.
- They turn blue litmus red.
- Strong acids have corrosive action on skin.
- Aqueous solutions of acids conduct electricity.
PROPERTIES OF BASES:
- Bases have bitter taste.
- They turn red litmus blue.
- Strong bases have corrosive action on skin.
- Aqueous solutions of bases conduct electricity.
2: State and Explain Arrhenius concept of acid and bases.
ARRHENIUS CONCEPT:
According to this concept, an acid is a substance that ionizes in water to produce H+ and a base is a substance that ionizes in water to produces OH– ions.
For example,
In the above examples HCl is an acid as it ionizes in water to produce H+ ion and NaOH is a base as it ionizes in water to give OH– ion.
What are the limitations of Arrhenius Concept?
LIMITATIONS:
- Arrhenius concept is only applicable in the presence of water.
- It can not explain why compounds such as CO2, SO2 etc., are acids and why substances like NH3, are bases as there is no H in CO2 and OH in NH3.
Write down uses of some important acids and bases
USES OF SOME IMPORTANT ACIDS:
- Hydrochloric acid, HCl, is used for cleaning of metals, bricks and removing scale from boilers.
- Nitric acid, HNO3, is used to manufacture of fertilizers, explosives.
- Sulphuric acid, H2SO4, is used to manufacture of many chemicals, drugs, dyes, paints and explosives.
- Phosphoric acid, H3PO4, is used to manufacture of fertilizers and acidulant for food.
USES OF SOME IMPORTANT BASES:
- Sodium hydroxide, NaOH, is used in soap making and drain cleaners.
- Potassium hydroxide, KOH, is used in making liquid soap and shaving cream.
- Calcium hydroxide, Ca(OH)2 is used in making mortar, plasters and cement.
- Magnesium hydroxide, Mg(OH)2, is used antacid and laxative.
State and Explain Lowrey and bronsted concept of acid and base
According to this theory an acid is a proton donor and a base is a proton acceptor.
Consider the following Example:
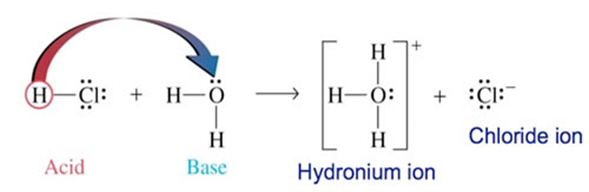
As HCl donates a proton to H2O so it is an acid. On the other hand as H2O accepts this proton so is a base.
Define Amphoteric Compounds?
Substances that react with both acids and bases are called amphoteric substances. For example, water
Above mentioned questions are also the important questions for chemistry class 10 chapter 2
We also provide mcqs for chemistry class 10 chapter 2
Notes of other chapter of class 10 chemistry
You can also download chemistry class 10 chapter 2 notes pdf